The concept of combustion. Conditions necessary for combustion to occur
The normal speed of flame propagation is the speed of movement of the flame front relative to unburned gas in a direction perpendicular to its surface.
The value of the normal speed of flame propagation should be used in calculating the rate of increase in the pressure of an explosion of gas and vapor mixtures in closed, leaky equipment and premises, the critical (extinguishing) diameter in the development and creation of flame arresters, the area of easily dropped structures, safety membranes and other depressurization devices; when developing measures to ensure the fire and explosion safety of technological processes in accordance with the requirements of GOST 12.1.004 and GOST 12.1.010.
The essence of the method for determining the normal speed of flame propagation is to prepare a combustible mixture of known composition inside the reaction vessel, ignite the mixture in the center with a point source, record the change in pressure in the vessel with time, and process the experimental pressure-time dependence using a mathematical model of the gas combustion process in closed vessel and optimization procedures. The mathematical model makes it possible to obtain a calculated dependence “pressure-time”, optimization of which according to a similar experimental dependence results in a change in the normal velocity during the development of an explosion for a particular test.
The normal burning rate is the rate at which the flame front propagates relative to the unburned reactants. The burning rate depends on a number of physicochemical properties of the reagents, in particular, thermal conductivity and the rate of a chemical reaction, and has a well-defined value for each fuel (under constant combustion conditions). In table. 1 shows the burning rates (and ignition limits) of some gaseous mixtures. Fuel concentrations in mixtures were determined at 25°C and normal atmospheric pressure. The flammability limits, with exceptions noted, were obtained with flame propagation in a 0.05 m diameter tube closed on both sides. The fuel excess coefficients are defined as the ratio of volumetric fuel contents in the real mixture to the stoichiometric mixture (j1) and to the mixture at the maximum burning rate (j2).
Table 1
Burning rates of condensed mixtures (inorganic oxidant + magnesium)
| Sheet |
| Document No. |
| Signature |
| the date |
| Sheet |
| TGIV 20.05.01.070000.000 PZ |
As can be seen, during the combustion of air gas mixtures at atmospheric pressure u max lies within 0.40-0.55 m/s, and - within 0.3-0.6 kg/(m2-s). Only for some low molecular weight unsaturated compounds and hydrogen u max lies within 0.8-3.0 m/s, and reaches 1-2 kg/(m2s). By magnification and max the studied fuels in mixtures with air can be
arrange in the following row: gasoline and liquid rocket fuels - paraffins and aromatics - carbon monoxide - cyclohexane and cyclopropane - ethylene - propylene oxide - ethylene oxide - acetylene - hydrogen.
| Change |
| Sheet |
| Document No. |
| Signature |
| the date |
| Sheet |
| TGIV 20.05.01.070000.000 PZ |
The linear combustion rate of oxygen mixtures is much higher than air mixtures (for hydrogen and carbon monoxide - 2-3 times, and for methane - more than an order of magnitude). The mass combustion rate of the studied oxygen mixtures (except for the CO + O2 mixture) lies in the range of 3.7–11.6 kg/(m2 s).
In table. Table 1 shows (according to the data of N. A. Silin and D. I. Postovsky) the burning rates of compacted mixtures of nitrates and perchlorates with magnesium. For the preparation of mixtures, powdered components were used with particle sizes of nitrates 150–250 μm, perchlorates 200–250 μm, and magnesium 75–105 μm. The mixture was filled into cardboard shells with a diameter of 24-46 mm to a compaction factor of 0.86. The samples were burned in air at normal pressure and initial temperature.
From a comparison of the data in Table. 1 and 1.25 it follows that condensed mixtures are superior to gas mixtures in terms of mass and are inferior to them in terms of linear burning rate. The burning rate of mixtures with perchlorates is less than the burning rate of mixtures with nitrates, and mixtures with alkali metal nitrates burn at a higher rate than mixtures with alkaline earth metal nitrates.
table 2
Flammability limits and burning rates of mixtures with air (I) and oxygen (II) at normal pressure and room temperature
| Sheet |
| Document No. |
| Signature |
| the date |
| Sheet |
| TGIV 20.05.01.070000.000 PZ |
| Change |
Methods for calculating the burnout rate of liquids
| Change |
| Sheet |
| Document No. |
| Signature |
| the date |
| Sheet |
| TGIV 20.05.01.070000.000 PZ |
![]() ; (16)
; (16)
where M is the dimensionless burnout rate;
 ; (17)
; (17)
M F- molecular weight of the liquid, kg mol -1 ;
d- characteristic size of the burning liquid mirror, m. It is determined as the square root of the combustion surface area; if the combustion area has the shape of a circle, then the characteristic size is equal to its diameter. When calculating the rate of turbulent combustion, one can take d= 10 m;
T to is the boiling point of the liquid, K.
The calculation procedure is as follows.
The combustion mode is determined by the value of the Galilean criterion Ga, calculated by the formula
where g- free fall acceleration, m·s -2 .
Depending on the combustion mode, the dimensionless burnout rate is calculated M. For laminar combustion mode:
For transient combustion mode:
if , then ![]() , (20)
, (20)
if , then , (21)
For turbulent combustion regime:
; ![]() , (22)
, (22)
M0- molecular weight of oxygen, kg mol -1 ;
n 0- stoichiometric coefficient of oxygen in the combustion reaction;
nF- stoichiometric coefficient of the liquid in the combustion reaction.
B- dimensionless parameter characterizing the intensity of mass transfer, calculated by the formula
![]() , (23)
, (23)
where Q- lower calorific value of liquid, kJ·kg -1 ;
| Change |
| Sheet |
| Document No. |
| Signature |
| the date |
| Sheet |
| TGIV 20.05.01.070000.000 PZ |
c- isobaric heat capacity of combustion products (assumed to be equal to the heat capacity of air c = 1), kJ·kg -1 ·K -1 ;
T0- ambient temperature, taken equal to 293 K;
H- heat of vaporization of the liquid at the boiling point, kJ·kg -1 ;
c e is the average isobaric heat capacity of the liquid in the range from T0 before T to.
If the kinematic viscosity of the vapor or the molecular weight and boiling point of the liquid under study are known, then the turbulent combustion rate is calculated using experimental data by the formula
where m i- experimental value of the burnout rate in the transient combustion mode, kg·m -2 ·s -1 ;
d i- the diameter of the burner in which the value is obtained m i, m. It is recommended to use a torch with a diameter of 30 mm. If a laminar combustion regime is observed in a burner with a diameter of 30 mm, a burner with a larger diameter should be used.
The normal speed of flame propagation (un) depends on the thermophysical properties of the gas-air mixture. But to an even greater extent, the speed of propagation depends on its physico-chemical properties? combustion rate V, and temperature in the combustion reaction zone, TG:
those. un is proportional to the rate of the oxidation reaction (V) and is exponentially dependent on the reciprocal temperature of the combustion zone (Tg). The determining parameter, of course, will be the speed of the reaction. We write the equation for the rate of the chemical reaction of combustion:

where k0 is the pre-exponential factor from the Arrhenius equation,
Cr, Juice - concentrations of fuel and oxidizer,
m, n - orders of the reaction, respectively, in fuel and oxidizer,
Ea is the activation energy of a chemical reaction.
Let us consider how the rate of oxidation reactions will change for mixtures with different ratios of fuel and oxidizer (Fig. 2).
It can be seen from the graph that for a mixture of stoichiometric composition (air excess coefficient? = 1), the rate of the oxidation reaction is maximum.
With an increase in the concentration of fuel in the mixture above the stoichiometric amount, when? becomes< 1 (кислород находится в недостатке), горючее сгорает не полностью. В этом случае меньше выделится теплоты реакции горения Qгор и произойдёт снижение Tг.
The rate of the oxidation reaction will decrease compared to the stoichiometric composition of the mixture, both due to a decrease in the concentration of the oxidizer O2 and the temperature of the combustion zone. That is, with a gradual decrease? (which is equivalent to an increase in the concentration of SG in the mixture) the rate of the oxidation reaction? and the temperature of the combustion zone Tg will consistently decrease. On the graph, at SG > SGstogh, the curve becomes sharply descending. The decrease in the rate of the oxidation reaction at? > 1 is explained by the decrease in heat release in the combustion zone due to the lower concentration of fuel in it.

Figure 2. The dependence of the burning rate on the concentration of fuel in the mixture
It is this dependence of the combustion reaction rate on the concentration of the combustible component in the initial mixture, as in Fig. 2, that predetermines the parabolic form of the dependence of its other parameters of the combustion process on the composition of the mixture: self-ignition temperature and minimum ignition energy, concentration limits of flame propagation. The dependence of the normal speed of flame propagation un on the concentration of fuel in the SG mixture also has the form of a parabola. On fig. Figure 3 shows such dependences for the case of combustion of an air-propane mixture at various values of the initial temperature.

Figure 3. Dependence of the flame propagation speed on the concentration of propane in the air at an initial temperature of 311 K (1); 644 K (2); 811K (3)
According to the concepts described above, the maximum flame propagation velocity (unmax) must correspond to the stoichiometric concentration of the fuel. However, its experimental values are somewhat shifted towards combustible mixtures rich in content. With an increase in the initial temperature of the mixture, the flame propagation velocity should increase, which is observed in practice. For example, for an air mixture of gasoline and kerosene vapors, it has the form shown in Fig. 4.

Figure 4. Dependence of the flame propagation speed on the initial temperature of the air mixture of gasoline and kerosene vapors with air
For various substances, un depends on their chemical nature and varies over a fairly wide range (Table 1). For most mixtures of hydrocarbon fuels with air< 1 м/с. При введении в горючую смесь избыточного воздуха или азота температура горения заметно снижается.
Table 1.
Normal flame propagation speed for some combustible mixtures
The introduction of inert and neutral gases into the combustible mixture: nitrogen N2, argon Ar, carbon dioxide CO2 dilutes it and thereby reduces both the rate of the oxidation reaction and the rate of flame propagation. This is clearly seen from the dependencies shown in Fig. 5.
At the same time, at a certain (phlegmatizing) concentration of diluents, combustion stops altogether. The introduction of freons has the strongest effect, since they also have an inhibitory effect on the combustion reaction.
As can be seen from fig. 5, the introduction of freon (114B2) into the combustible mixture is 4 - 10 times more effective than neutral gases - diluents.

Figure 5. Influence of the concentration of diluents and freon 114V2 on the flame propagation speed in the propane-air mixture (? = 1.15)
The phlegmatizing ability of gases - diluents depends on their thermophysical properties, and in particular, on their thermal conductivity and heat capacity.
1.3 Diffusion combustion of gases
In real conditions, in cases where gas or vapors ignite after the start of their emergency outflow, diffusion combustion is observed. A typical and fairly common example is the diffusion combustion of gas during the destruction of main pipelines, at an emergency gushing offshore or onshore well of a gas or gas condensate field, at gas processing plants.
Let us consider the features of such combustion. Let's assume that a fountain of natural gas is burning, the main component of which is methane. Combustion occurs in the diffusion mode and has a laminar character. The concentration limits of flame propagation (KPRP) for methane are 5 - 15% vol. Let us depict the structure of the flame and plot the graphic dependences of the change in the methane concentration and the rate of the combustion reaction on the distance to the axial fountain (Fig. 6).

Figure 6. Scheme of a diffusion laminar flame of a gas fountain (a), change in fuel concentration (b), combustion reaction rate (c) along the flame front.
The gas concentration decreases from 100% at the axial fountain to the value of the upper concentration ignition limit and further to the LEL at its periphery.
The combustion of the gas will occur only in the concentration range from VKPR to LEL, i.e. within the concentration region of its ignition. The combustion reaction rate? (T) will be equal to zero at concentrations above the VCR and below the LEL, and maximum at. Thus, the distance between HNKPR and HVKPR determines the width of the diffusion flame front:
fp = HNKPR - HVKPR. (3)
The front width for such a flame ranges from 0.1 to 10 mm. The rate of the combustion reaction in this case is determined by the rate of diffusion of oxygen, and its value is about 5 × 104 times less than the rate of combustion in the kinetic regime. The heat density is lower by the same factor, i.e. the rate of heat release in a diffusion-burning torch.
1.4 Peculiarities of combustion of gas jets. Flame Stabilization Conditions
It is more convenient to consider the combustion conditions of gas fountains using the example of gas jets. Under real conditions, such jets are turbulent. When a gas jet flowing from a well is ignited, a so-called diffusion flame is formed, which has a symmetrical spindle shape (Fig. 6). Chemical combustion reactions take place in a thin surface layer of the flame, which, in the first approximation, can be considered a surface where the concentrations of fuel and oxidizer vanish, and the diffusion flows of fuel and oxidizer to this surface are in a stoichiometric ratio. The diffusion combustion front has a zero propagation velocity, so it cannot stay on its own on the upward flowing jet.
Stabilization of the flame on the jet occurs in the lowest part of the torch, where a different combustion mechanism is realized. When gas flows out of the hole, a turbulent layer of mixing of gas and ambient air is formed on the initial non-burning section of the jet surface. In this layer, the gas concentration gradually decreases in the radial direction, while the concentration of the oxidizer increases. In the middle part of the mixing layer, a homogeneous mixture of fuel and oxidizer with a composition close to stoichiometric arises. When such a mixture prepared for combustion is ignited, the flame front can propagate in the mixing layer at a finite velocity even towards the flow if the combustion velocity exceeds the local flow velocity. But since the jet velocity increases as it approaches the outlet, then at a certain height the jet velocity (uf) becomes equal to the combustion velocity (?t), and the flame stabilizes on the jet surface at this height. It is not possible to accurately calculate the turbulent combustion rate (?t). However, estimates show that the value (?t) is approximately equal to the pulsating jet velocities, the value of which is proportional to the axial velocity (um). It follows from the experimental data that the maximum values of the root-mean-square fluctuations of the longitudinal velocity component are 0.2um. Taking this value as the speed of turbulent combustion, we can assume that the maximum speed of flame propagation towards the gas jet spouting at a speed of 300-450 m/s will be about 50 m/s.
1.5 Estimation of the flow rate of burning gas fountains
When extinguishing fires of powerful gas fountains, it becomes necessary to assess the flow rate (D) of a burning fountain, since gas consumption is one of the main parameters that determine the amount of work and material and technical means necessary to eliminate the accident. However, direct measurement of the flow rate of a burning fountain in most cases is impossible, and there are no effective remote methods for determining the flow rate of the jet. The flow rate of powerful gas fountains can be determined quite accurately from the height of the torch (H).
It is known that the height of a turbulent flame formed during the combustion of normally expanded gas jets with a subsonic outflow velocity does not depend on the speed or flow rate of the jet, but is determined only by the diameter of the hole (d) from which the jet flows, the thermophysical properties of the gas and its temperature (T) at the outlet of the hole.
There is a well-known empirical formula for calculating the flow rate of a fountain according to the height of the torch during the combustion of natural gas:
D = 0.0025Hf 2, million m3/day. (4)
In real fires, the laminar regime of combustion is practically not found. Gas, both in the reservoir of a gas field, and in transport pipelines and in technological installations, is under pressure. Therefore, the gas consumption in case of an emergency outflow will be very large? up to 100 m3/s on fires of flowing gas wells (up to 10 million m3/day). Naturally, under these conditions, the outflow regimes, and hence the combustion regimes, will be turbulent.
To calculate the forces and means to extinguish burning gas torches, it is necessary to know the gas flow rate. The initial data for its calculation are almost always absent, since either the gas pressure in the process equipment or in the reservoir reservoir is unknown. Therefore, in practice, they use the experimentally established dependence (4) of the flame height of the torch on the gas flow rate, the calculated data using which are given in Table. 2.
Table 2.
The dependence of the flame height on the gas flow rate of the gas fountain for various combustion modes
Combustion- these are intense chemical oxidative reactions, which are accompanied by the release of heat and luminescence. Combustion occurs in the presence of a combustible substance, an oxidizing agent and an ignition source. Oxygen and nitric acid can act as oxidizing agents in the combustion process. As fuel - many organic compounds, sulfur, hydrogen sulfide, pyrite, most metals in free form, carbon monoxide, hydrogen, etc.
In a real fire, the oxidizing agent in the combustion process is usually atmospheric oxygen. The external manifestation of combustion is a flame, which is characterized by luminescence and heat release. When burning systems consisting only of solid or liquid phases or their mixtures, a flame may not occur, i.e., occurs flameless burning or smoldering.
Depending on the state of aggregation of the initial substance and combustion products, homogeneous combustion, combustion of explosives, and heterogeneous combustion are distinguished.
Homogeneous combustion. In homogeneous combustion, the initial substances and combustion products are in the same state of aggregation. This type includes the combustion of gas mixtures (natural gas, hydrogen, etc. with an oxidizing agent, usually air oxygen) /
Burning explosives associated with the transition of a substance from a condensed state to a gas.
heterogeneous combustion. In heterogeneous combustion, the initial substances (for example, solid or liquid fuel and gaseous oxidizer) are in different states of aggregation. The most important technological processes of heterogeneous combustion are the combustion of coal, metals, the combustion of liquid fuels in oil furnaces, internal combustion engines, combustion chambers of rocket engines.
The movement of a flame through a gas mixture is called flame spread. Depending on the speed of propagation of the combustion flame, it can be deflagration at a speed of several m/s, explosive at a speed of the order of tens and hundreds of m/s, and detonation at thousands of m/s.
Deflagration combustion is subdivided into laminar and turbulent.
Laminar combustion is characterized by a normal flame propagation velocity.
Normal flame propagation speed called the speed of movement of the flame front relative to unburned gas, in a direction perpendicular to its surface.
Temperature increases the normal speed of flame propagation relatively little, inert impurities reduce it, and an increase in pressure leads either to an increase or decrease in the speed.
In a laminar gas flow, the gas velocities are small. The burning rate in this case depends on the rate of formation of the combustible mixture. In a turbulent flame, the swirling of gas jets improves the mixing of the reacting gases, since the surface through which molecular diffusion occurs increases.
Indicators of fire and explosion hazard of gases. Their characteristics and scope
The fire hazard of technological processes is largely determined by the physical and chemical properties of raw materials, intermediate and final products circulating in the production.
Fire and explosion hazard indicators are used in the categorization of premises and buildings, in the development of systems to ensure fire safety and explosion safety.
Gases are substances whose absolute vapor pressure at a temperature of 50 °C is equal to or greater than 300 kPa or whose critical temperature is less than 50 °C.
For gases, the following values apply:
Flammability group- an indicator that is applicable to all aggregate states.
Flammability is the ability of a substance or material to burn. According to the combustibility of substances and materials are divided into three groups.
non-combustible(fireproof) - substances and materials that are incapable of combustion in air. Non-combustible substances can be flammable (for example, oxidizing agents, as well as substances that release combustible products when interacting with water, atmospheric oxygen, or with each other).
slow-burning(flammable) - substances and materials that can ignite in the air from an ignition source, but are not able to burn on their own after its removal.
combustible(combustible) - substances and materials capable of spontaneous combustion, as well as ignite from an ignition source and burn independently after its removal. Flammable substances and materials are distinguished from the group of combustible substances and materials.
Flammable substances and materials that can ignite from a short-term (up to 30 s) exposure to a low-energy ignition source (match flame, spark, smoldering cigarette, etc.) are called flammable.
The combustibility of gases is determined indirectly: a gas that has concentration limits of ignition in air is referred to as fuel; if the gas does not have concentration limits of ignition, but ignites spontaneously at a certain temperature, it is classified as slow-burning; in the absence of concentration limits of ignition and autoignition temperature, the gas is classified as non-combustible.
In practice, the combustibility group is used to subdivide materials by combustibility, when establishing classes of explosive and fire hazardous zones according to the PUE, when determining the category of premises and buildings according to explosion and fire hazard, and when developing measures to ensure fire and explosion safety of equipment and premises.
Auto ignition temperature- the lowest temperature of a substance at which, under the conditions of special tests, there is a sharp increase in the rate of exothermic reactions, ending in fiery combustion.
The concentration limits of flame propagation (ignition) - that the range of concentrations in which combustion of mixtures of combustible vapors and gases with air or oxygen is possible.
Lower (upper) concentration limit of flame propagation - the minimum (maximum) content of fuel in a mixture of combustible substance-oxidizing medium "at which flame propagation through the mixture is possible at any distance from the ignition source. Within these limits, the mixture is combustible, and outside of them, the mixture is unable to burn.
Temperature Limits of Flame Propagation(ignition) - such temperatures of a substance at which its saturated vapors form in a particular oxidizing environment concentrations equal, respectively, to the lower (lower temperature limit) and upper (upper temperature limit) concentration limits of flame propagation.
The ability to explode and burn when interacting with water, atmospheric oxygen and other substances- a qualitative indicator that characterizes the special fire hazard of certain substances. This property of substances is used when determining the category of production, as well as when choosing safe conditions for conducting technological processes and conditions for joint storage and transportation of substances and materials.
Laminar combustion rate is the speed with which the flame front moves in the direction perpendicular to the fresh FA surface.
 – zone of laminar combustion;
– zone of laminar combustion;
 is the rate of laminar combustion.
is the rate of laminar combustion.
turbulent combustion.

Turbulent flame speed is the speed at which the flame front moves in a turbulent flow.
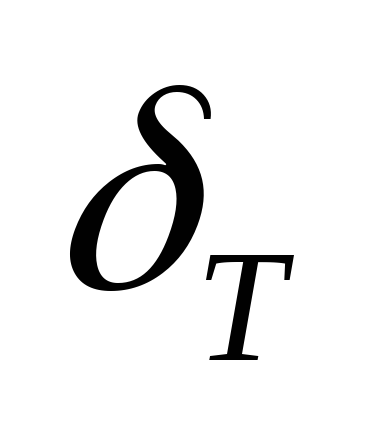 – zone of turbulent combustion;
– zone of turbulent combustion;

 are the normal velocities of small particles.
are the normal velocities of small particles.
Laminar combustion does not provide the required rate of heat release in the engine, so turbulence of the gas flow is required.
Arrhenius equation:
 is the rate of a chemical reaction.
is the rate of a chemical reaction.
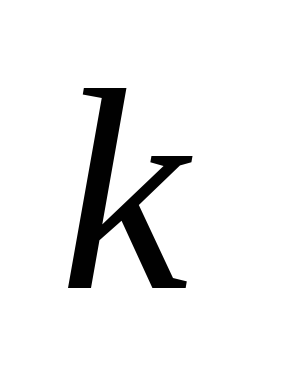 is the constant of the chemical reaction, depending on the composition of the mixture and the type of fuel;
is the constant of the chemical reaction, depending on the composition of the mixture and the type of fuel;
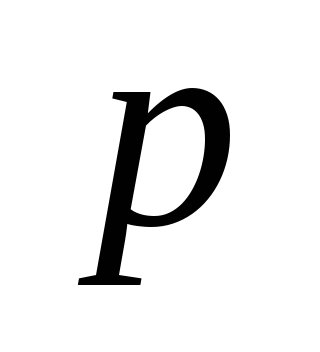 is the pressure of a chemical reaction;
is the pressure of a chemical reaction;
 – the order of a chemical reaction;
– the order of a chemical reaction; 
 is the universal gas constant;
is the universal gas constant;
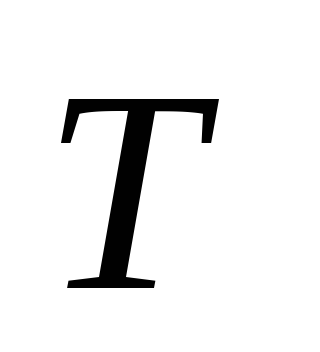 is the temperature of the chemical reaction;
is the temperature of the chemical reaction;
 -activation energy - the energy required to break intramolecular bonds.
-activation energy - the energy required to break intramolecular bonds.
Influence of various factors on the combustion process in internal combustion engines with spark ignition.
The composition of the mixture.
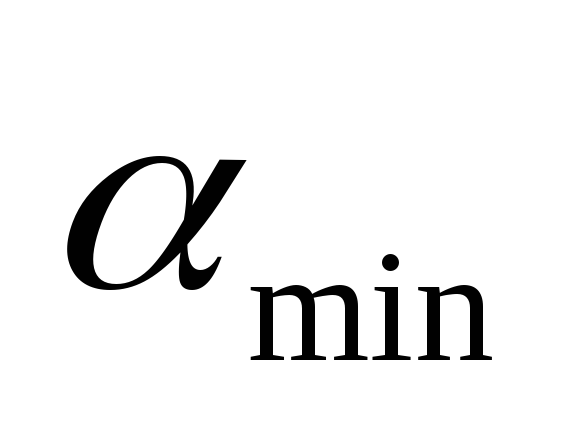
 – upper concentration limit;
– upper concentration limit;
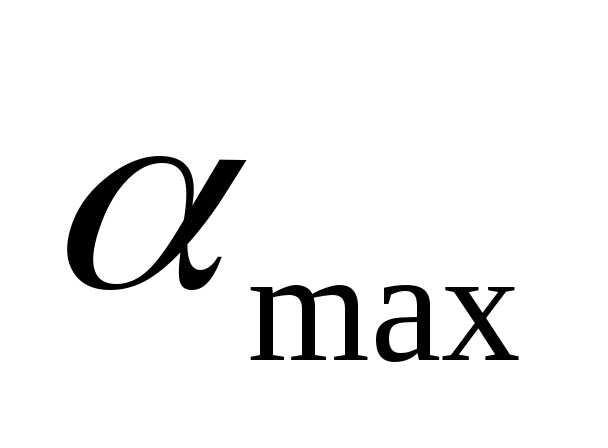 –lower concentration limit;
–lower concentration limit;
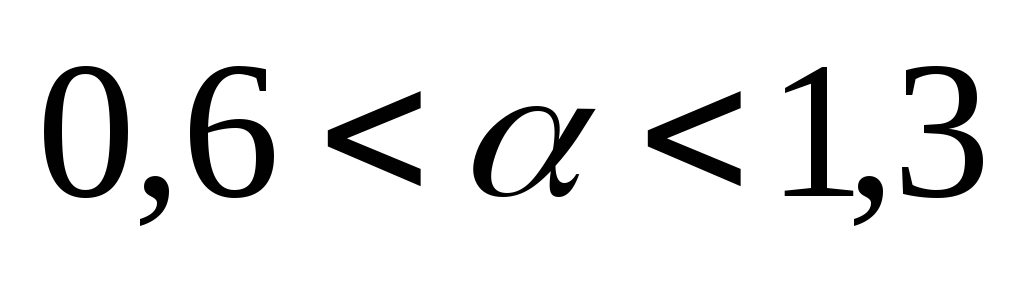 – normal combustion;
– normal combustion;
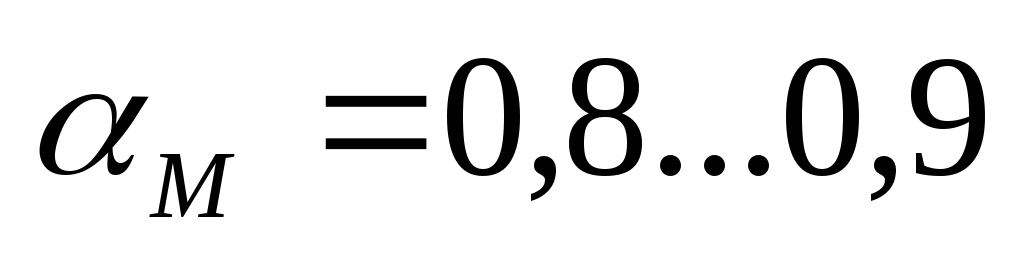
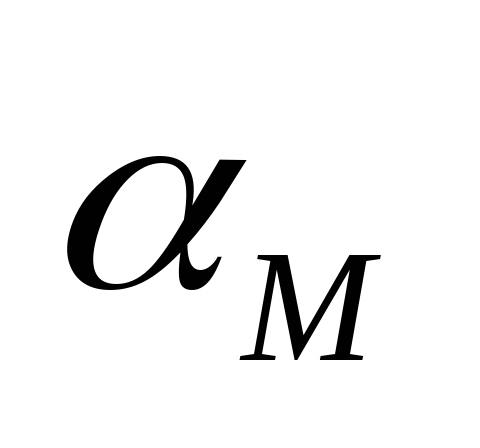 –power composition of the mixture
- the maximum power developed by the engine.
–power composition of the mixture
- the maximum power developed by the engine.
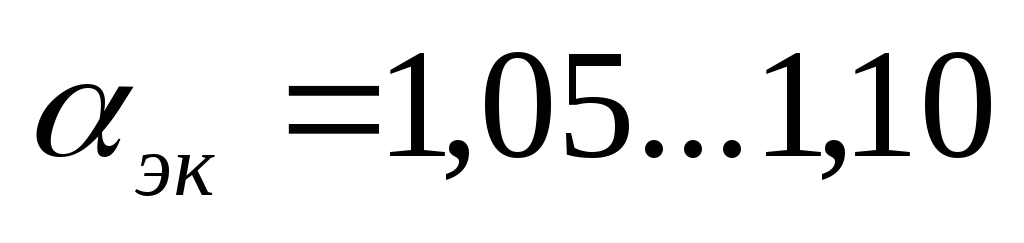
 –economic composition of the mixture
- maximum economy.
–economic composition of the mixture
- maximum economy.
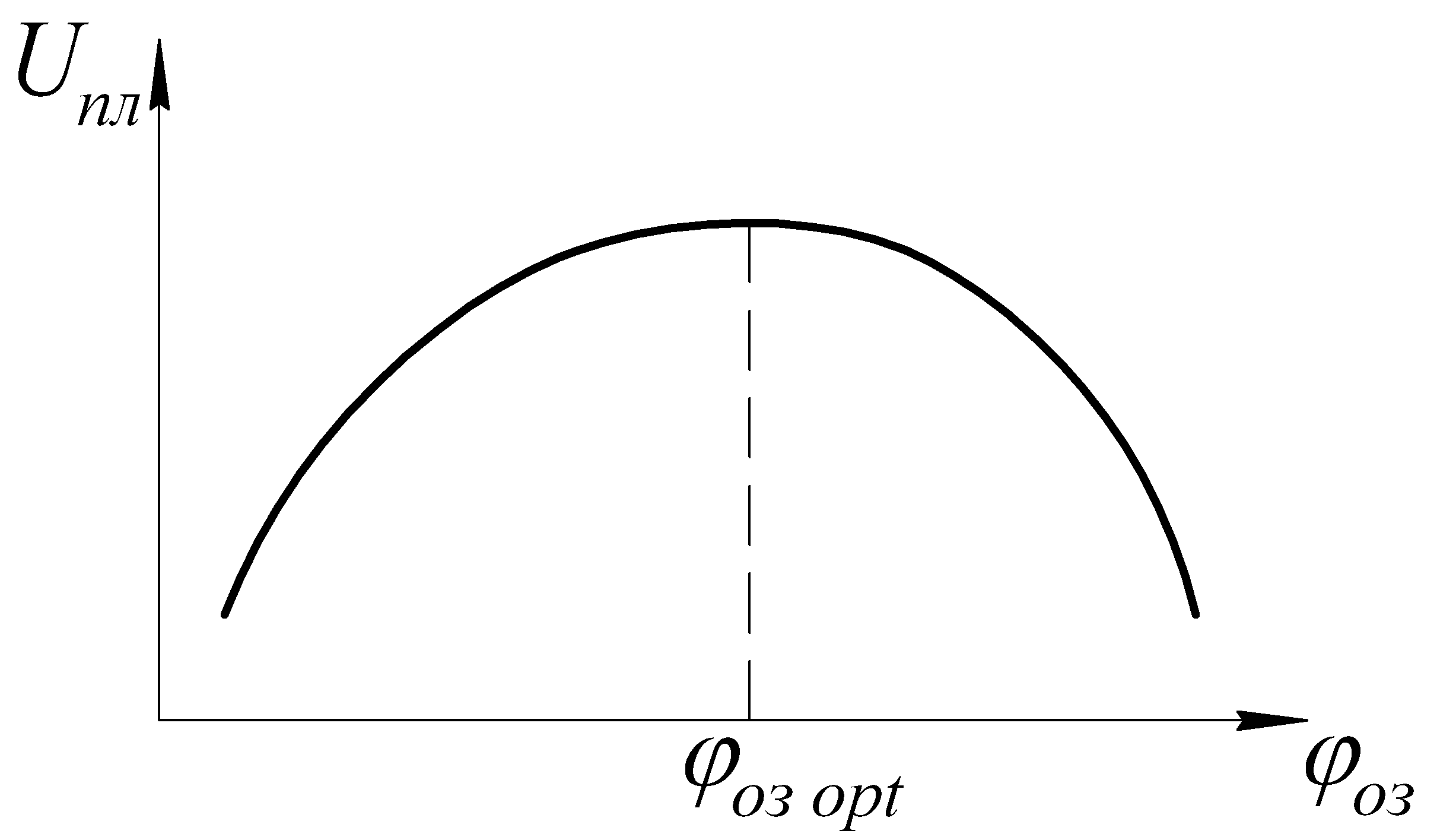
Compression ratio.



With an increase in the frequency of revolutions, the ignition phase increases, which leads to a late development of the combustion process and a decrease in the amount of heat released per cycle. Therefore, when changing  Ignition advance adjustment (IUZ) is required.
Ignition advance adjustment (IUZ) is required.
Ignition advance angle.
Ignition advance angle - the angle of rotation of the crankshaft from the moment the spark is applied to TDC.
P  one load
understand the angle of rotation of the throttle - it is she who regulates the load on the engine.
one load
understand the angle of rotation of the throttle - it is she who regulates the load on the engine.
 - Throttle angle.
- Throttle angle.
The main violations of the combustion process in internal combustion engines with spark ignition. Detonation.
D  contonation
- explosive combustion of the mixture, accompanied by pressure shock waves propagating throughout the volume of the combustion chamber. Detonation occurs as a result of self-ignition of parts of the mixture remote from the candle, due to intense heating and compression during the propagation of the flame front.
contonation
- explosive combustion of the mixture, accompanied by pressure shock waves propagating throughout the volume of the combustion chamber. Detonation occurs as a result of self-ignition of parts of the mixture remote from the candle, due to intense heating and compression during the propagation of the flame front.

On detonation: 
Reflecting from the walls of the combustion chamber, the shock wave forms secondary flame fronts and self-ignition centers. Externally, detonation manifests itself in the form of dull knocks when the engine is running at high loads.
Consequences of the operation of the engine with detonation:
Overheating and burnout of individual engine components (valves, pistons, head gaskets, spark plug electrodes);
Mechanical destruction of engine parts due to shock loads;
Reduced power and efficiency.
That. prolonged operation with detonation is unacceptable.
P  Here are the factors that cause detonation:
Here are the factors that cause detonation:

The ability of a fuel to self-ignite characterizes detonation resistance , and the detonation resistance is estimated octane number (OC) .
OC – numerically equal to the volume fraction of a mixture of poorly detonating isooctane with easily detonating normal heptane, equivalent in detonation properties to this gasoline.
Isooctane - 100 units, normal heptane - 0 units.
For example: a 92 octane rating indicates that this gasoline has the same knock resistance as a reference blend of 92% isooctane and 8% normal heptane.
BUT  – automobile gasoline;
– automobile gasoline;
and - research method for obtaining gasoline;
m - motor method (the letter is usually not written).
In the motor research method, the compression ratio is adjusted until detonation begins, and the octane number is determined from the tables.
motor methods simulate driving at full load (truck outside the city).
research method simulates movement at partial load (in the city).


If the octane number is excessively high, then the speed of flame propagation is reduced. The combustion process is delayed, which leads to a decrease in efficiency and an increase in the temperature of the exhaust gases. The consequence of this is a drop in power, an increase in fuel consumption, overheating of the engine and burnout of individual elements. The maximum performance of the engine is achieved when the octane number of the fuel is close to the detonation threshold.
Ways to deal with detonation:

the distance traveled by the flame front per unit time. (See: ST SEV 383-87. Fire safety in construction. Terms and definitions.)
Source: "House: Building terminology", Moscow: Buk-press, 2006.
- A measure of the prevalence of a disease based on its prevalence in a population, either at a point in time) or over a specified period of time)...
medical terms
- - Movement of the flame root zone from the burner outlets in the direction of fuel or combustible mixture flow See all terms of GOST 17356-89. BURNERS ON GAS AND LIQUID FUELS...
Dictionary of GOST vocabulary
- - Displacement of the root zone of the flame towards the outflowing mixture See all terms of GOST 17356-89. BURNERS FOR GAS AND LIQUID FUELS. TERMS AND DEFINITIONS Source: GOST 17356-89...
Dictionary of GOST vocabulary
- - Alternating change in the torch parameters and localization of its root zone See all terms of GOST 17356-89. BURNERS FOR GAS AND LIQUID FUELS. TERMS AND DEFINITIONS Source: GOST 17356-89...
Dictionary of GOST vocabulary
- - a phenomenon characterized by the escape of the flame into the body of the burner. Source: "House: Building terminology", M.: Buk-press, 2006...
Construction dictionary
- - propagation of fiery combustion over the surface of substances and materials. Source: "House: Building terminology", M.: Buk-press, 2006...
Construction dictionary
- - the degree of duration of transportation of goods by rail ...
Reference commercial dictionary
- - hemodynamic indicator: the speed of movement of the pressure wave caused by the systole of the heart along the aorta and large arteries ...
Big Medical Dictionary
- - a device that detects a flame and signals its presence. It may consist of a flame detector, an amplifier and a relay for signal transmission...
Construction dictionary
- - a phenomenon characterized by a general or partial detachment of the base of the flame above the burner openings or above the flame stabilization zone. Source: "House: Building terminology", M.: Buk-press, 2006...
Construction dictionary
- - one of the physical properties of coal, measured by objective quantitative methods. Closely related not only to the structure and composition, but also to the presence of cracks and pores, as well as miner. impurities...
Geological Encyclopedia
- - velocity of propagation of the elastic perturbation phase in dec. elastic environments. In unbounded isotropic media, elastic waves propagate adiabatically, without dispersion...
Geological Encyclopedia
- - "... - a conditional dimensionless indicator characterizing the ability of materials to ignite, spread flame over the surface and generate heat ..." Source: "FIRE SAFETY REGULATIONS ...
Official terminology
- - "...: an indicator characterizing the ability of a paintwork to ignite, spread a flame over its surface and generate heat ..." Source: "SAFETY OF PAINT AND VARNISH MATERIALS ...
Official terminology
- - FLAMES. Flame, etc. see the flame...
Explanatory Dictionary of Ushakov
- - adj., number of synonyms: 2 smoldering smoldering ...
Synonym dictionary
"flame speed" in books
Ice and some fire
From the book On all four sides author Gill Adrian AnthonyIce and a little fire Iceland, March 2000 Why, with such an abundance of God-created lands, did anyone come here at all? And why, having come here and looked around, these people did not turn their family boat around and sail away to hell with all their children and
twin flames
From the book Soul Integration by Rachel SalTwin Flames Greetings dear ones, this is Leah. Again, it gives me great pleasure to speak with you. All the time that the Arcturians, the Founders and the Higher Self of this channel have communicated with you, we have also been with you. Now we will talk about a topic close to our hearts
DEDICATED TO THE FLAME
From the book Mystery of Fire. Collection author Hall Manly PalmerDEDICATED TO THE FLAME He who lives Life will know
1.6. Can the speed of information exchange exceed the speed of light?
From the book Quantum Magic author Doronin Sergey Ivanovich1.6. Can the speed of information exchange exceed the speed of light? Quite often one hears that experiments to test Bell's inequalities, which refute local realism, confirm the presence of superluminal signals. This means that information can
Meditation on the flame
From the book of Wisdom. Mantras. Meditations. Key Practices by Loy-SoFlame Meditation There is another type of meditation that has a powerful healing and healing effect. This is candle meditation. The flame has long been revered in all cultures, as well as the ashes, representing the purified essence of the subject. It was believed that
UPR. Meditation on the flame
From the book NOTHING ORDINARY author Millman DanUPR. Flame Meditation The next time you have unpleasant, disturbing thoughts, do a simple but powerful meditation: Take a steady and steady burning candle. Place it on a table away from flammable objects such as curtains.
Speed of propagation of gravitational interactions
From the book Gravity [From crystal spheres to wormholes] author Petrov Alexander NikolaevichVelocity of Propagation of Gravitational Interactions At the end of the chapter, we will discuss one more interesting problem. General relativity includes two fundamental constants: gravitational G and the speed of light c. The presence of the first of them is obvious and natural - we are dealing with
19.22. Extinguishing the flame
From the book of Stratagems. About the Chinese art of living and surviving. TT. 12 author von Senger Harro19.22. Extinguishing the Flame As long as the Arabs were successful in the Yom Kippur War (October 6-22, 1973), the Soviet Union did not demand a ceasefire. October 9 at
Propagation speed
From the book Daily Life of Medieval Monks of Western Europe (X-XV centuries) author Moulin LeoSpeed of spread The breadth of spread is remarkable, but even more impressive is the speed with which the influence of monasticism spread. For as soon as it became known that a handful of people settled in some "desert", as literally right there around them
In flames
From the book Partisans accept battle author Lobanok Vladimir EliseevichIn flames The war has left a deep, indelible mark on everyone who survived it. Events bother her every day, it happens that they don’t let her sleep at night, the wounds of her heart that still haven’t cooled disturb her. So it probably should be, it will still be as long as those who were at the front are alive
LECTURE XI THREE WAYS OF DISTRIBUTION OF MAGNETIC INFLUENCE. – 1) MENTAL PHOTOGRAPHY. – 2) SOLAR PLEXUS METHOD. – 3) MUSCULAR WAY THREE WAYS OF DIRECT DISTRIBUTION OF MAGNETIC INFLUENCE.
From the book Personal Magnetism (lecture course) author Daniels Van TailleLECTURE XI THREE WAYS OF DISTRIBUTION OF MAGNETIC INFLUENCE. – 1) MENTAL PHOTOGRAPHY. – 2) SOLAR PLEXUS METHOD. – 3) MUSCULAR WAY THREE WAYS OF DIRECT DISTRIBUTION OF MAGNETIC INFLUENCE. When applying each of the three methods, it is necessary first of all
Lesson 1. Sts. apostles from 70: Jason, Sosipater and other holy martyrs with them (On what the holy apostles did to spread the Christian faith and what we must do to spread it)
From the book Full Yearly Circle of Brief Teachings. Volume II (April–June) author Dyachenko Grigory MikhailovichLesson 1. Sts. apostles from 70: Jason, Sosipater and other holy martyrs with them (On what the holy apostles did to spread the Christian faith and what we must do to spread it) I. Sts. Apostles Jason and Sosipater, whose memory is being celebrated today, disciples and
The speed of practice reading should be three times the speed of normal reading.
From the book Speed Reading. How to remember more by reading 8 times faster by Kamp PeterPractice reading speed should be three times faster than normal reading. The basic rule of practice is that if you want to read at a certain speed, then you need to do practice reading about three times faster. So,
52. Water hammer wave velocity
From the book Hydraulics author Babaev M A52. Velocity of propagation of a hydraulic shock wave In hydraulic calculations, the velocity of propagation of a shock wave of a hydraulic shock, as well as the hydraulic shock itself, is of considerable interest. How to define it? To do this, consider a round transverse
51. Outflow velocity in a narrowing channel, mass flow velocity
From the book Heat Engineering author Burkhanova Natalia51. Outflow velocity in a narrowing channel, mass flow velocity Outflow velocity in a narrowing channel Let's consider the process of adiabatic outflow of matter. Let us assume that a working fluid with a certain specific volume (v1) is in a reservoir under
What else to read
THE LAST NOTES
- Amber extraction method in Belarus
- “Soon products from amber will arrive on store shelves”: trial amber mining has begun in the Brest region
- How to open an IP in Belarus
- Is it necessary to pay contributions to the social security budget for an employee who is on unpaid leave?
- What to give a man after parting
